Studying membrane proteins is not easy. The broad scope of the problem clearly deserved a Noble prize in 2012. Thanks to these advances, today scientists can determine structures of some membrane proteins (e.g., G protein-coupled receptors). But some of them are so huge and complex that X-Ray crystallography and NMR spectroscopy don’t help. Continue reading “Nanopharmacology: [atomic] force awakens”
Month: December 2015
Acute and chronic neuroscience
Those who know even a tiny bit of toxicology, are well aware about difference between acute and chronic effects of xenobiotics. Some chemicals (or other factors, for that matter) seem to be harmless at the first touch (acute) but tend to develop terrible effects if taken continuously over long time (chronic). The common sense explanation for that is accumulation of small damage over time in our inner organs (liver, for instance), although it could be a misconception. Continue reading “Acute and chronic neuroscience”
Retrosynthesis ex machina
A Christmas present from one of the bastions of modern organic synthesis appeared in Nature. The idea of automated retrosynthetic analysis bugged the greatest OC minds since EJ Corey came up with the very concept of retrosynthesis. Back in 1969 Corey and Wipke set the stage by publishing a paper in Science.

OC tidbits #1
Despite deromantizing total synthesis I do like organic chemistry and do feel aesthetic pleasure from synthetic schemes and mechanisms [note for my future employer, I do enjoy the bench work, too!]. Also, since high school and undergrad years I still have weak spot for org-chem puzzles. So Below are some little nuggets (subjective, of course) from the recent OC papers that I found interesting.
-
From Popov et al (Somfai lab) (ACIE):
- Try to figure out the mechanism
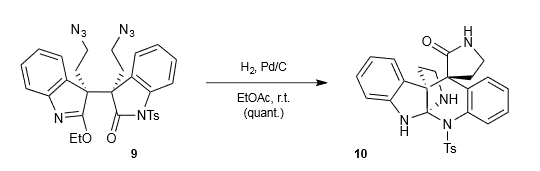
- Try to explain the difference in chemoselectivity (and what’s going on with that Ts group by the way?).


-
From Hamasaki et al (Kochi lab) (JACS)
Can you guess the mechanism for this one?

-
Dream big!
All multi-ton processes start from the small scale, check this cute Kugelrohr apparatus setup by Thiyagarajan et al. for processing of biomass-derived furans (The white stuff in the middle is a zeolite catalyst; ACIE)

Synthetic lethality as drug discovery platform
One of the features of drug design in the -omics era is the shift from target- and structure-based to function-based drug discovery, when the active compound is identified simultaneously or before the mechanism of action.
A new report in Nature Chemical Biology describes an interesting blend of small molecule high-throughput screening with genetic screening via synthetic lethality. As one might guess, the approach is dealing with cellular death. Traditional ‘simple’ genetic screen identifies individual genes that are critical for cell survival. The principle of synthetic lethality is somewhat different. Scientists seek gene pairs or networks that are crucial in combination but which could be silenced individually without jeopardizing essential cellular functions. Previously it was applied for the discovery of anticancer therapeutics. This time the team from Harvard Medical School aimed at Staphyllococcus aureus. Continue reading “Synthetic lethality as drug discovery platform”
Antiviral sugar superballs

It’s not too common to encounter a compound as one above in a paper tagged ‘medicinal chemistry’. But the team of Spanish and French chemists seems not to care too much about rule of 5 or any other rules of thumb that constrain their creativity. Especially when the rationale for compound design was:
it is difficult to make compounds of an adequate size and multivalency to mimic natural systems such as viruses
The ligands dubbed ‘superballs’ are targeting dendritic cell-specific intercellular adhesion molecule-3-grabbing non-integrin (DC-SIGN). And the multivalency of ligand-receptor interaction is the key factor of viral infection. Previous state-of-the-art ligands were virus-like particles with 1620 mannoses displayed on their surface. So I guess in this study authors significantly optimized ligand efficiency. Continue reading “Antiviral sugar superballs”
What’s so special about synthesis?
On Tuesday a piece form Phillip Ball appeared in Nature, with provocative question ‘Why synthesize?’ addressed to organic chemists, and particularly to adepts of total synthesis. And, you know what, after reading it, I didn’t feel like the author answered the question − for each argument he provides a counterargument. So the take home message I got was the following:
even though molecule-building is sure to remain a crucial part of the chemical enterprise, conventional organic synthesis need not be the only, or even the best, way to do it
When I started learning about organic chemistry, the book “Organic synthesis: the science behind the art” was one of the strongest inspiration sources for the subsequent choice of the major. This perception of the organic synthesis as the art is something that triggered my interest but what’s causing awkward feelings nowadays.
Like architecture, chemistry deals in elegance in both design and execution.
What exactly is meant by “elegance” of synthesis? “Beauty is in the eyes of the beholder” says the old proverb. So isn’t it disturbing that organic chemists themselves are the only ones who appreciate the elegance of total synthesis?
In my current opinion arranging synthetic steps in a reasonable manner is an engineering and project management problems, but by no means is it the art. There’s no more elegance in designing the synthetic scheme than in designing any other sophisticated multi-step experiment. So why don’t call microbiology the art, too? Why setting up the culturing of stubborn microorganisms producing natural products is different? After all, it’s often the same trial and error process yield a couple milligrams of the product in the end.
For me the synthesis is a means, not the goal. It’s a chemist’s way of solving problems. But other scientists may be interested in solving the same problems, too. So indeed, the synthesis is not the only and often not the best way to do it. That’s why chemists should focus on how to make it the best way. In this I’m staying with George Whitesides’s philosophy that simplicity should be the major goal. But that doesn’t make organic chemists unique among other scientists. That’s why I’d rephrase the following quote from Phillip Ball:
We need to avoid romanticizing an imagined bygone age […]
We [organic chemists] need to avoid romanticizing an imagined uniqueness of our field and train a broader look on problem solving.

Writing code for synthetic life
In the high school my chemistry teacher used to tell us that chemists do not only study the nature, but they also invent their own subject of study. The more I learn about biology, the more I feel that biologists move in the same direction, creating the new field of synthetic biology.
Recent report on the expansion of DNA alphabet by two letters grossly overshadowed not that press-release-friendly development in the synthetic biology of RNA. But there are quite some interesting things going on in the latter field that deserve as much attention. Continue reading “Writing code for synthetic life”
Rational serendipity for drug design
With all modern advances and super-sexy scientific tools we currently have, randomness is still arguably the best source of truly innovative discoveries. And scientists do acknowledge that, so various “accelerated serendipity” techniques are flourishing in drug discovery, organic chemistry, chemical biology, and other fields. Continue reading “Rational serendipity for drug design”
Expecting price drop for Americium?
This paper caught my attention immediately as I was scrolling through the rss feed. It’s not that I’m generally interested in chemistry of actinide metals. But for a chemist it’s always refreshing to be reminded of elements from the subscript of the periodic table. Continue reading “Expecting price drop for Americium?”
![Nanopharmacology: [atomic] force awakens](https://bernatv.files.wordpress.com/2015/12/sert.png?w=661)

